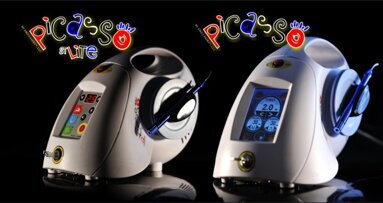
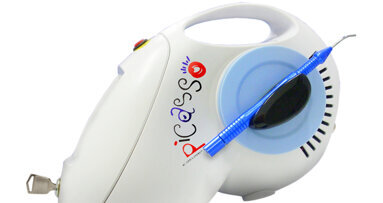
NEW YORK, NY, USA: With international certifications now in place for its Picasso diode laser, AMD LASERS is poised for continued growth in North America as well as expansion into the European market.
AMD LASERS, based in Indianapolis, IN, USA, has attained International Organization for Standardization (ISO) 13485:2003 certification of its medical device quality management system in North America, Canada and the European Union. In addition, the quality management system demonstrates conformance to the Medical Device Directive 93/42/EEC, allowing the products to be CE marked, which makes them available to be sold in the European Economic Area.
“Achieving ISO 13485 certification further demonstrates our leadership and commitment to the highest standards of our industry,” said Alan Miller, president and CEO of AMD LASERS, in a news release announcing the certification. “We also have many potential customers in Europe eager to purchase the Picasso, so our ability to use the CE mark is another major step forward in our mission to provide advanced, affordable dental lasers around the world. It’s a proud day for our young company.”
ISO 13485:2003 is the internationally recognized standard for the development, production, and servicing of medical products. In order to obtain ISO 13485 certification, a company must demonstrate the ability to provide medical devices and related services that consistently meet customer and regulatory requirements applicable to medical devices and related services.
During the certification process, AMD LASERS partnered with TUV Rheinland, an international service group that serves as an ISO registrar, auditing the safety and quality of new and existing products, systems and services.
“It has been a pleasure to work with TUV Rheinland,” said Amy Szentes, compliance manager at AMD LASERS. “They are the gold standard in ISO registrars, and we feel this certification validates all of the hard work we have put into both product quality and compliance.”
ANAHEIM, CA, USA: AMD LASERS, the company that builds the world’s most affordable dental laser technology, announces the launch of the Picasso soft ...
NEW YORK, NY, USA: For any dentist interested in adding laser technology to his or her practice, AMD LASERS, a company based in Indianapolis, IN, USA, has ...
NEW YORK, NY, USA/LEIPZIG, Germany: The US-based manufacturer AMD LASERS recently launched the Picasso Lite in dental markets worldwide. As a first in the ...
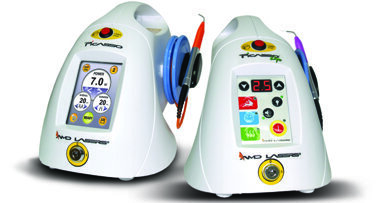
TULSA, OKLA., USA: AMD LASERS, a global developer and manufacturer of dental lasers and provider of dental laser education, has announced that its Picasso ...
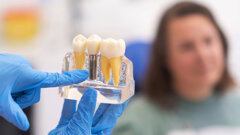
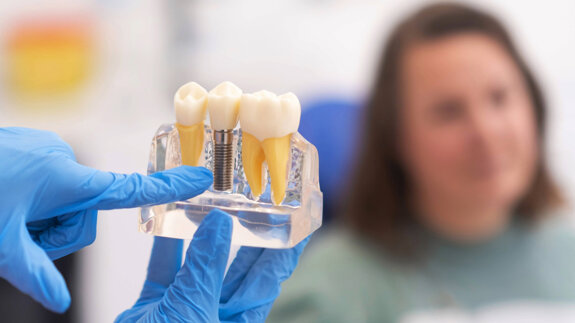
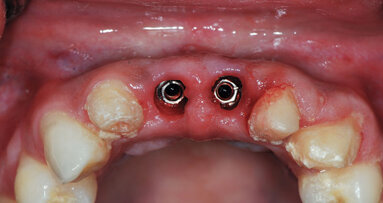
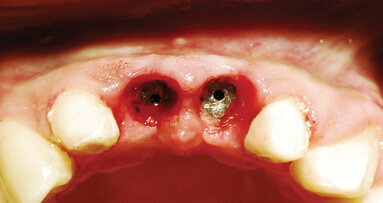
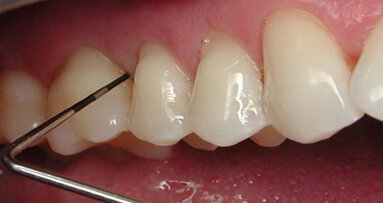
Dental implants are placed either utilizing a one-stage approach (healing abutment placed at implant placement) or a two-stage approach (implant is covered ...
AMD LASERS — a company based in Indianapolis, IN, USA, that manufactures laser therapy equipment for dentists that is easy to use and affordable ...
AMD LASERS, the Indianapolis-based company that makes using lasers affordable and accessible to dentists, has introduced a new option for its Picasso and ...
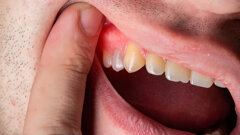
The concept of using dental lasers for the treatment of periodontal disease elicits very strong reactions from both ends of the spectrum. Everyone has an ...
Dental implants are placed either utilizing a one-stage approach (healing abutment placed at implant placement) or a two-stage approach (implant is covered ...
While dental lasers have been commercially available for several decades and their popularity among patients is unparalleled, the dental profession has ...
Live webinar
Tue. 14 April 2026
8:00 PM EST (New York)
Dr. Bruce McFarlane Certified Specialist in Orthodontics Fellow: Royal College of Dentists of Canada Diplomate: American Board of Orthodontics
Live webinar
Wed. 15 April 2026
2:00 PM EST (New York)
Live webinar
Wed. 15 April 2026
8:00 PM EST (New York)
Linda Hecker MS, BSDH, RDH
Live webinar
Thu. 16 April 2026
12:00 PM EST (New York)
Dr. Pär-Olov Östman, Dr. Robert Gottlander DDS
Live webinar
Fri. 17 April 2026
12:00 PM EST (New York)
Live webinar
Fri. 17 April 2026
1:00 PM EST (New York)
Dr. Stuart Yeaton BDentSc, MOrth
Live webinar
Mon. 20 April 2026
1:00 PM EST (New York)
Dr. Alberto Monje DDS, MS, PhD



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 International / International
International / International
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East













































To post a reply please login or register