WASHINGTON, DC, USA: A newly-tested treatment for multiple sclerosis reduces relapse and deterioration in patients, without many of the negative side effects associated with other treatments. A clinical study published Wednesday in the New England Journal of Medicine (NEJM) said that Cladribine, which is produced by pharmaceutical group Merck Serono, was the first oral medication for patients suffering from multiple sclerosis, a complex neurological condition.
A team led by Gavin Giovanonni, a professor at Barts and The London School of Medicine and Dentistry in Britain, followed some 1,300 patients for nearly two years, monitoring them with MRI scans.Patients were either given two or four short courses of Cladribine tablets over the course of a year or were administered a placebo. The results were heartening, with those taking the Cladribine tablets over 55 percent less likely to suffer relapse and 30 percent less likely to suffer worsening in their disability due to multiple sclerosis.
The disease is a neurological condition that often begins in young adulthood and involves the body's immune system attacking its own central nervous system. The damage done interferes with communications between the brain and other parts of the body leading to disability.
Patients are often required to undergo regular injections and intravenous infusions, but Cladribine, which works by suppressing the immune system, only needs to be taken for between eight to 10 days a year to work effectively, the study found.
"The introduction of an oral therapy, particularly one that has no short-term side effects and is as easy to use as oral Cladribine, will have a major impact on the treatment of multiple sclerosis," Giovanonni said:
Merck Serono's request to put the drug on the market was rejected by the US Food and Drug Administration at the beginning of December. It is now in discussions with the US regulatory body and seeking to resubmit its application, according to a statement from the company.
Multiple sclerosis usually affects people between 20 and 40 years old, and its symptoms can be serious and crippling. It can affect almost all of the biological functions that control movement, sensory perception and memory.
An estimated one in 1,000 people are affected by the disease.
We hear about it a lot these days — dental practices getting squeezed by lower consumer spending. The result is less treatment acceptance and lower ...
As dental professionals, we have all had that gut-wrenching moment when a cancer patient walks through our door mid-treatment. Their mouths are full of ...
Vancouver/New York, USA: Dentists might find it awkward to treat patients who are in wheelchairs. For those who can’t be transferred into a dental ...
NEW YORK, N.Y., USA: Dental implant surgery procedures can conflict with the medication needs of heart patients. A less invasive approach to dental implants...
FREMONT, Calif., US: For many orthodontists, obtaining optimal tooth alignment and managing final detailing are persistent challenges in aligner therapy. ...
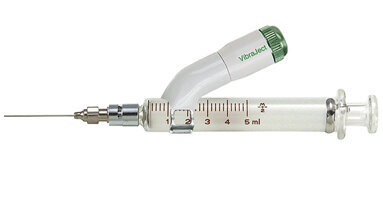
The VibraJect multiple-use Dental Needle Accessory is unique in that it can be effectively utilized for endodontic irrigation by merely attaching it to the ...
SureSmile digital orthodontics delivers a comprehensive, integrated solution designed to support diagnostic precision, workflow efficiency and predictable ...
NEW YORK, N.Y., USA: Cardiac patients who take anticoagulant medications and need a tooth extraction face an increased risk of bleeding that must be ...
LOUISVILLE, Ky., USA: During a check-up at the Kentucky Racing Health Services Center (KRHSC), one patient is diagnosed with an abscessed tooth and another ...
CHICAGO, Ill., USA: The American Dental Association (ADA) commends Congress for passing the Action for Dental Health Act. For years, dentists have worked to...
Live webinar
Wed. 3 June 2026
1:00 PM EST (New York)
Live webinar
Thu. 4 June 2026
2:00 PM EST (New York)
Live webinar
Mon. 8 June 2026
12:00 PM EST (New York)
Live webinar
Mon. 8 June 2026
1:00 PM EST (New York)
Dr. Anthony Mak B.D.S, Prof. Marleen Peumans
Live webinar
Mon. 8 June 2026
2:00 PM EST (New York)
Live webinar
Wed. 10 June 2026
11:00 AM EST (New York)
Live webinar
Wed. 10 June 2026
2:00 PM EST (New York)
Nacho Fernández-Baca DDS, MSc



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 International / International
International / International
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East












































To post a reply please login or register