Platelet-rich fibrin can play an important role in oral and maxillofacial surgery, implant dentistry, periodontal regeneration and post-extraction site preservation. The fibrin is a reservoir of platelets that will slowly release growth factors and cytokines, which are the key factors for regeneration of the bone and maturation of the soft tissue. Platelet-rich fibrin (PRF) is an autologous platelet concentrate prepared from the patient’s own blood at the dentist’s office just before the oral/dental procedure.
Recent studies are focused on the development of natural therapeutic alternatives, which are easy to prepare, non-toxic or biocompatible to living tissues and economically inexpensive. The goal is the local release of growth factors, in turn accelerating hard- and soft-tissue healing.
PRF is a natural fibrin-based biomaterial prepared without anticoagulants or artificial additives (biomedical modifiers) that allow us to obtain autologous fibrin membranes and plugs with a high concentration of platelets and white cells, releasing growth factors at the surgical site for seven to 14 days and accelerating the natural healing process.
Evidence from literature suggests the potential role PRF provides in most oral-surgery procedures is key for regeneration and tissue engineering. The slow polymerization during centrifugation and fibrin-based structure could make PRF a better healing biomaterial than PRP and other fibrin adhesives, but literature is also controversial, with many publications demonstrating excellent results with PRP as well as with PRF.
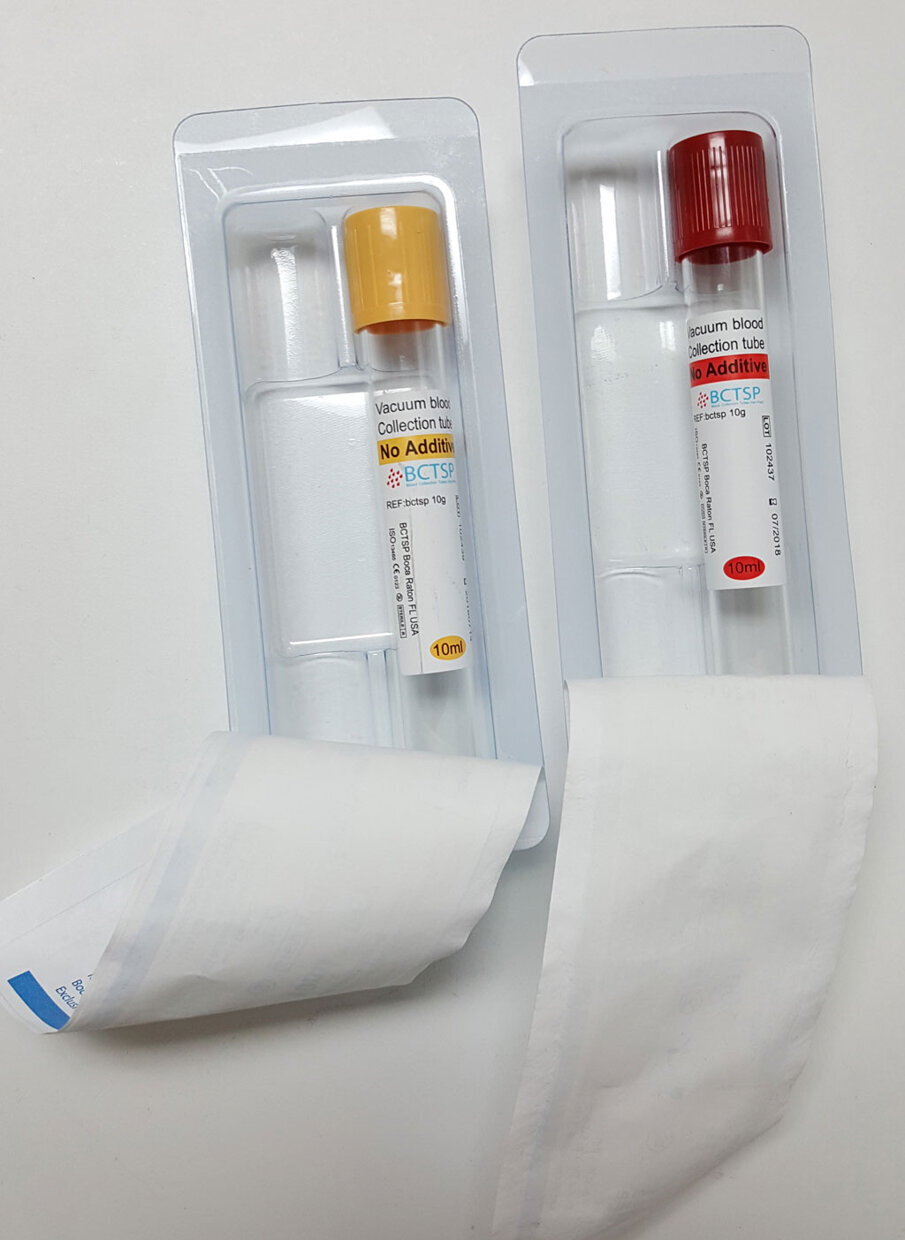
Fig. 1: Boca Dental Supply blood collection tubes steri-pack are single-use, medical-grade packages for blood collection and manufacturing of PRF. Discard any unused tubes.
Fig. 2: Doctor usually expends less than two minutes drawing patient’s blood.
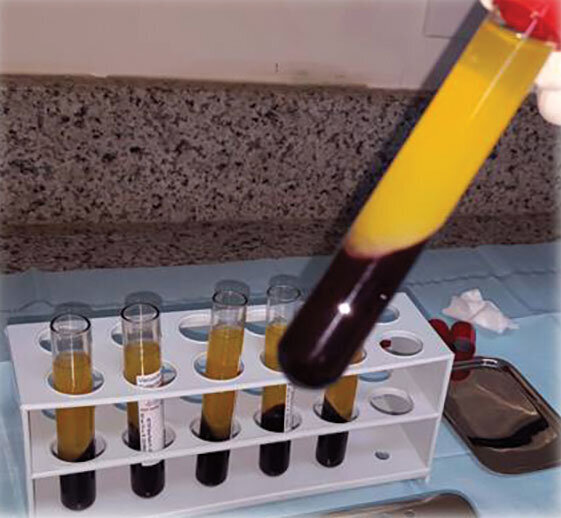
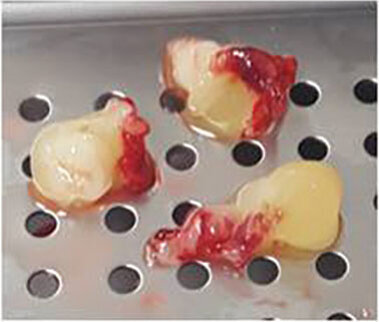
Fig. 3: PRF clots in the PRF box.
Fig. 7: Plain natural glass tube vs. silica coated tube.
Fig. 8: Plain natural glass tube vs. silica coated tube.
Fig. 9: Plain natural glass tube vs. silica coated tube.
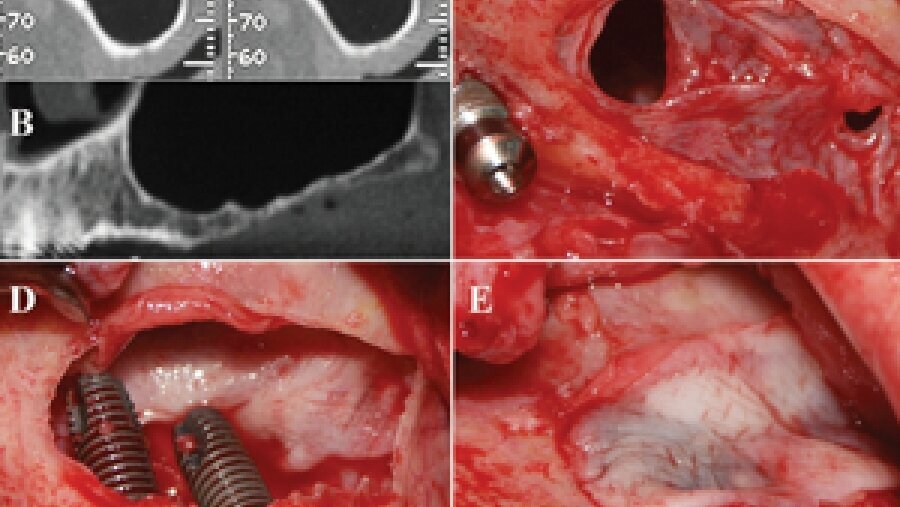
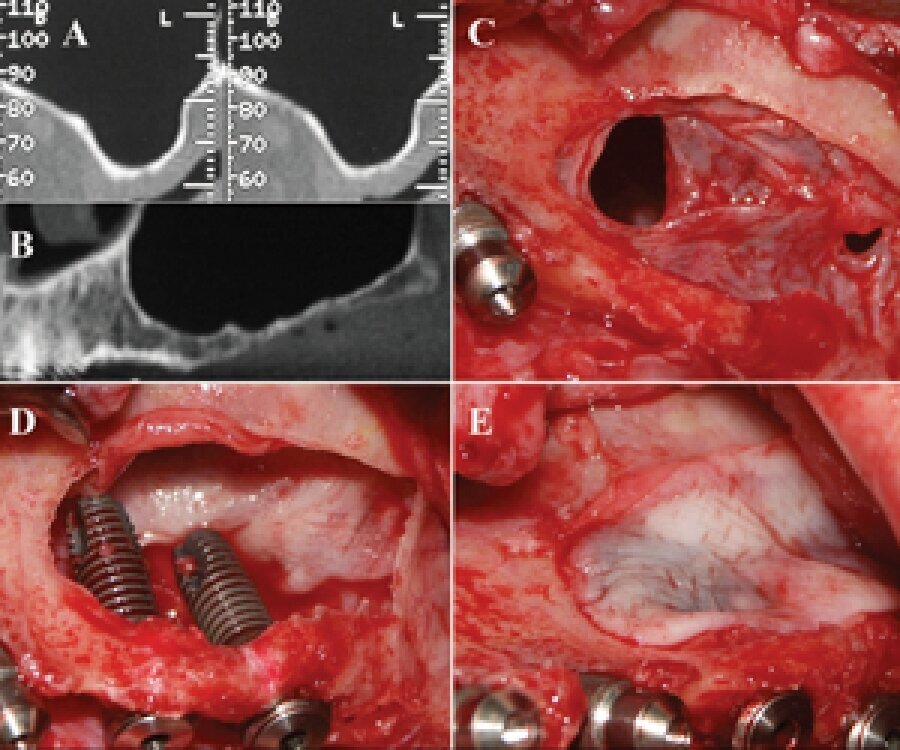
Figs. 10a-e: PRF is used in invasive surgery.
There is evidence that the presence of growth factors and cytokines in platelets play key roles in inflammation and wound healing. Platelets also secrete fibrin, fibronectin and vitronectin, which act as a matrix for the connective tissue and as adhesion molecules for more efficient cell migration. This has led to the idea of using platelets as therapeutic tools to improve tissue repair during wound healing.
Because of the benefits to soft tissue, PRP is now being used all over the word for facial rejuvenation, joint regeneration, hair growth stimulation and ED treatment. And, because of the easier and less expensive alternative, PRF liquid is starting to be used instead of PRP (PRP $450 vs. PRF $8 per patient).
Advantages of PRF compared with PRP
A look at the advantages of PRF as compared wtih PRP:
- No anticoagulants that affect the release of growth factors.
- No drugs (calcium chloride) that could affect fibrin polymerization.
- No animal products (Bovine thrombin) that could affect the coagulation process and immune system activation.
- PRF has the presence of natural fibrin network, which protects the growth factors from proteolysis.
- PRF favors the development of micro vascularization leading to a more efficient cell migration.
- PRF has the presence of monocytes, leukocytes and other white cells that have an important role during the inflammatory phase of healing.
- PRF manufacturing requires minimum time from the doctor.
The manufacturing of all blood concentrates at the patient’s site of treatment brings new challenges to the dentists and staff members: Infection-control protocols, staff training, education and research of the products used during PRF manufacturing.
Handling patient’s blood and manufacturing blood products transforms the dental office into a blood bank facility where stricter cross-contamination control protocols should be followed in order to avoid doctor’s liability risks and to comply with federal regulations of the Center for Disease Control (CDC), OSHA and to perform at the standard of care protocols for surgery.
PRF is used in invasive osseous surgery close to the eyes, ear, brain and in direct contact with bone, maxillary sinus, veins, arteries and nerves that could be adversely affected, if proper contamination control protocols are not followed.
All instruments used for the manufacturing of PRF should be sealed sterile and dropped into a sterile field separate from the instruments used for the removal of contaminated tissues, debridement of bone and teeth extraction. Two fields protocol will eliminate the risk of contamination of the PRF membranes, PRF sticky and PRF steaky bone that is going to be used for bone augmentation, as well as the PRF exudate that can be used as a sealant of the surgical site.
Tourniquets, bandages, gauze, needles and blood collection tubes should be single-patient-use packs only. I use the blood collection tubes steri-pack (BCTSP) from Boca Dental Supply, LLC.
BCTSP tubes are the only single-use, medical-grade packages for blood collection and manufacture of PRF existing in the market globally. After a tubes pack is open, any unused tubes should be discarded.
The manufacturing of PRF membranes and plugs is a simple, four-step protocol:
- Venipuncture for blood collection using needles and glass tubes from single-use packs only (medical-grade packs).
- Use the centrifuge to obtain separation and clot of the blood components.
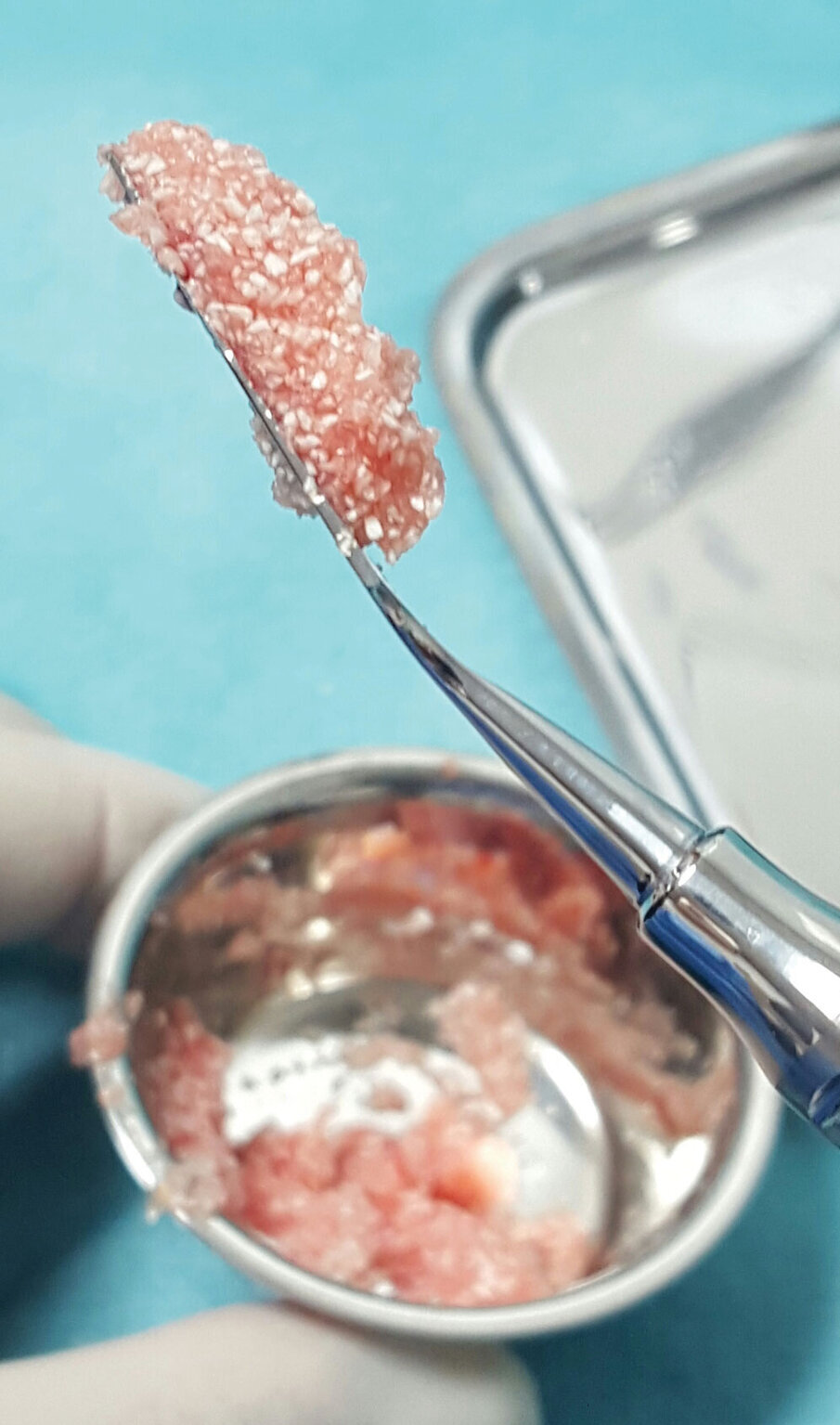
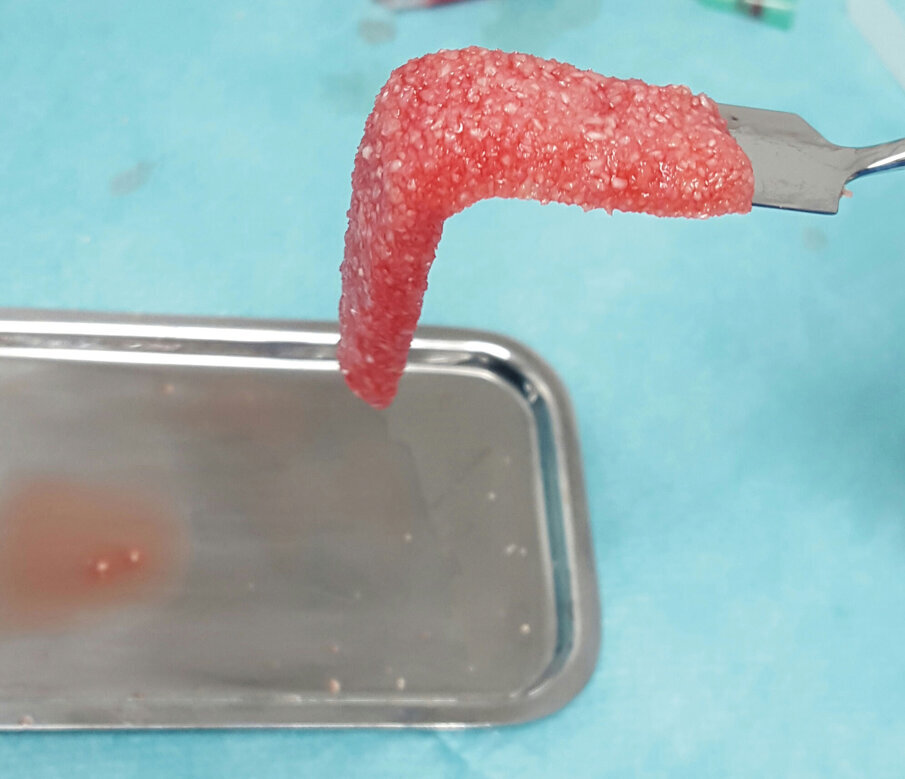
- Obtain the PRF clot from the tube and process it in the PRF box to produce membranes or plugs.
- Use membranes and plugs to mix with bone.
The doctor usually expends less than two minutes drawing blood, and the rest of the procedure to manufacture PRF can be performed by a properly trained staff member.
How is PRF clot formed?
After the blood is collected into the glass tubes and during the eight-minute centrifugation, the contact of blood coagulation factors with the natural hydrophilic glass surfaces activates the clotting cascade leading to the conversion of fibrinogen to fibrin forming a natural PRF clot.
If plastic tubes were going to be used for PRF clot, PRF membranes and PRF plugs, such tubes would likely have additives like silica and other dangerous chemical products to simulate the clothing characteristics of the natural glass, and the final product would be a chemically induced artificial PRF clot that will produce artificial PRF membranes and plugs.
The use of plastic tubes with silica coating and other chemicals to simulate the natural characteristics of the glass brings the challenge of not knowing what kind of damage the dentist would be causing to the patient’s health. The literature and research evidence has shown that silica and other coating with chemicals or additives used in laboratory blood-collection tubes increase the risk of cancer and damage the DNA. The use of plastic tubes silica and other hidden additives could be detrimental and contradictory to the basic philosophy of PRF when it was adapted from cardiovascular and general surgery to dentistry: “No anticoagulants and no additives.”
More research is needed to determine the final damage of silica and other additives in the plastic blood-collection tubes to the grafted area and grafted bone at post-extraction sites, maxillary sinus, periodontal defects and all other bone-
augmentation procedures. There is currently not available publication or research to evaluate possible cancer and systemic effects of silica and all other chemicals used to simulate the natural glass in plastic laboratory tubes when used for PRF manufacturing.
When plastic blood collection tubes without any additives are used for blood collection and centrifugation, we obtain liquid PRF that is used to apply to the sticky bone and transform it into PRF steaky bone. This improves the handling characteristics of the bone and aids in keeping the bone-graft material in solid form and preventing small particles of bone from migrating between the patient’s bone and periosteum. Migration of small particles of bone could be a cause of increased inflammatory response and swelling after surgery.
Because the time in the centrifuge is reduced to process blood in the plastic tubes to manufacture PRF liquid, less heat will be generated thus allowing a greater number of live white cells without degradation. This will accelerate the healing process; and it is also possible that when the blood is processed at 700 RPM or less, some stem cells could also be concentrated in the PRF liquid.
PRF is the newest and most popular technique to accelerate healing in dentistry. During most large implant dental conventions and meetings in oral and maxillofacial surgery, periodontics, OMS, endodontics, implantology and bone regeneration, the number of speakers presenting successful cases increases every year.
We, as clinicians involved in regenerative procedures and the manufacturing of PRF, are obligated to use only materials and supplies that guarantee patients’ safety and, at the same time, eliminate the clinician’s liability risks.
Note: Dr. Alvaro Betancur is the inventor of the Blood Collection Tubes Steri-Pack (BCTSP).
Tags:



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 International / International
International / International
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East













































Do dentists normally test for HIV or other blood borne infections as part of the PRF process or is the fact that single use instruments, gloves etc. mean that testing for such isn’t done as part of the PRF protocol?