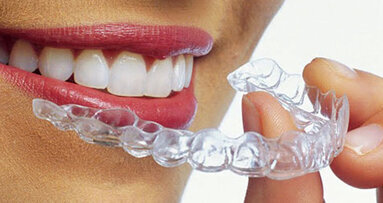
BREA, Calif., USA: Ormco Corp. has received FDA clearance to treat mixed dentition with its Spark Clear Aligner System, enabling orthodontists to treat younger patients.
According to a company press release, the clearance by the FDA makes Spark Clear Aligners one of a few doctor-directed aligners cleared in the U.S. for the treatment of younger patients.
The Release 11 features new innovations and an improved user experience including new anatomical beveled attachments for patient comfort, redesigned bite ramps that enable doctors to customize for each specific tooth type to enhance engagement between the bite ramps and the teeth and downloadable STL files to offer doctors in-office solutions, the company said.
“With more than 75 percent of orthodontic case starts each year being kids and teens, we are excited that we can now enable doctors to offer the same innovative benefits of Spark treatment to younger patients,” said Eric Conley, president of Spark Clear Aligners and Digital Orthodontics. “We are also confident the advancements of this new release will solidify Spark as the aligner of choice for orthodontists seeking great clinical outcomes and providing industry leading comfort and clarity for their patients.”
Release 11 key features enable improved clinical outcomes, more predictable and efficient treatment planning and greater patient comfort, the company said. The features include the following:
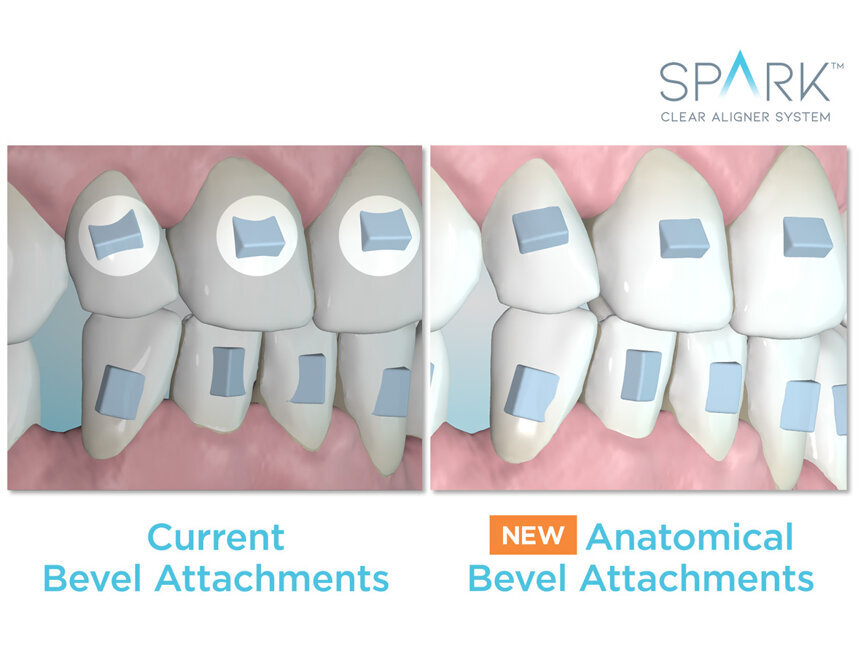
Anatomical beveled attachments conform to the surface of each tooth to provide a more uniform active attachment surface and to minimize sharp edges for patient comfort (data on file at Ormco).
“This enhances the product’s appeal to the patient,” said Dr. Bill Dischinger, a consultant of Ormco. “You already have the best-performing plastic, clearest plastic, most stain-resistant plastic and more comfortable edges on the market. Now, Ormco has enhanced the attachment comfort; you have the whole package.”
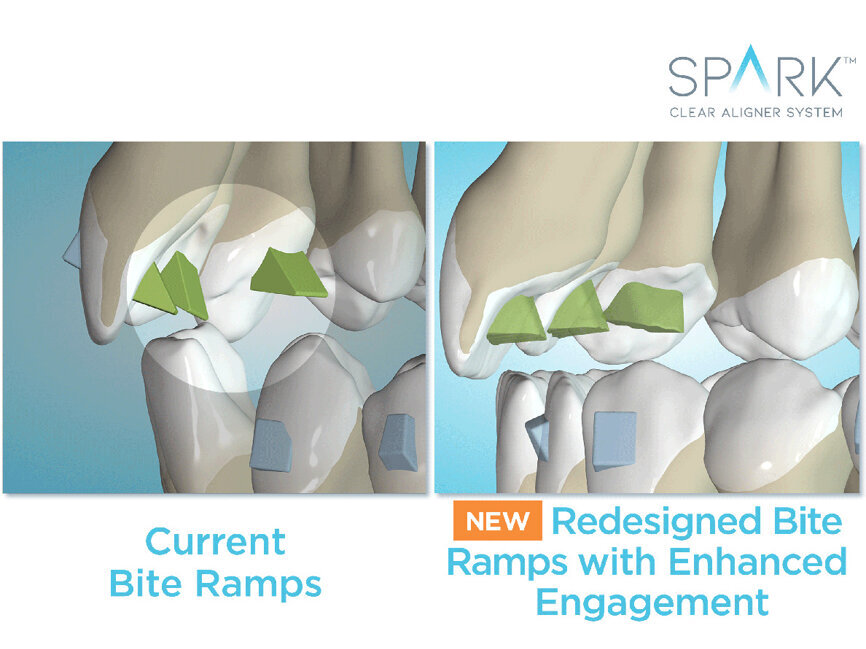
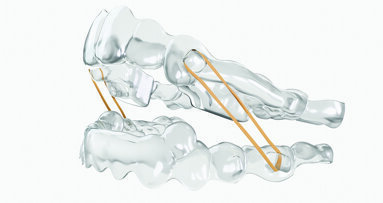
Redesigned bite ramps enhance the engagement between the bite ramps and the teeth and enable doctors to customize bite ramps for each specific tooth (incisors, canines, premolars) so that the active surface is oriented parallel to the occlusal plane. The new bite ramps have been lab tested for strength and durability (data on file at Ormco).
Dr. Trevor Nichols, a consultant of Ormco, said, “Deep bite cases are known for their difficulty. Paralleled bite ramps will offer more predictable intrusive movement as they direct the force down the long axis of the tooth and will create less undesired tipping and friction points within the aligner leading to more efficient and predictable intrusion.”
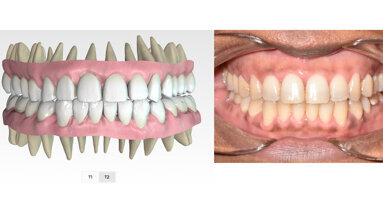
Mixed detention support in the Approver software allows doctors to treat younger patients (kids and teens) with mixed dentition, including providing eruption guides for semi-erupted teeth and numbering primary teeth. “This is a significant step to creating a Phase I/Phase II aligner approach to cases,” Nichols said. “Not only will Approver software enable doctors to better treat interceptive cases, but eruption guides will help control the eruption of the permanent dentition.”
Downloadable STL files of the first three stages and the last stage are now available for doctors to download from the Spark portal.
Nichols said, “With the ability to print the starter aligners, doctors can get a jump start on their cases and increase their case load and profitability. The last stage aligner allows the doctor and patient to have final models to use for retention for years to come.”
Dr. Nicole Scheffler, consultant of Ormco, added, “This really is a game-changer for patients, doctors and treatment coordinators.”
Spark Approver software enhancements
The Spark Release 11 includes the following Approver software enhancements:
- Continued Evolution of CBCT integration based on doctor input. Spark Approver now features several new improvements for more seamless and effective integration of CBCT data for Spark Aligners. Additional diagnostic tools include a 2-D clipping plane featuring multiple views, new tooth identification options, and enhanced CBCT performance and visual rendering.
- An enhanced tooth movement table now includes both crown and root movement values, as well as the addition of both tip and torque movement for intermediate and challenging movement.
- User interface improvements based on doctor input include a new preset for overjet, a new panel docking/undocking feature, redesigned and more friendly menu options, and simpler dialog boxes.
Enhancements for easier case management
- New packaging improvements require less storage by including more aligners per box and reduces packaging waste. The introduction of connected aligner bags and Spark Aligners’ packaging inserts will help ensure that aligner bags remain in order.
- More efficient workflow through staff accounts allows orthodontic team members the ability to submit and manage Spark Aligner patients through the Spark Portal and Approver software. Support for multiple billing addresses for practices that have multiple offices will be available in August 2021.
More information on the Spark Aligner product and software enhancements is available at ormco.com/Spark.
(Source: Ormco)
Tags:
Ormco Corp., a global leader in orthodontic solutions, has announced the launch of its Spark On-Demand program, which enables doctors to order any number of...
Ormco Corp., a leading provider of innovative orthodontic solutions for more than 60 years, is excited to announce the official launch of Spark Retainers ...
Brea, Calif. (Aug. 8, 2023) – Ormco Corporation announces the highly anticipated Spark Clear Aligners Release 14. Release 14 continues Spark’s ...
Swiss manufacturer Dentalpoint AG, a supplier of metal-free oral rehabilitation products, recently announced the U.S. arrival of what it describes as the ...
ORANGE, Calif., USA: Ormco Corp., a manufacturer and provider of orthodontic technology and services, has announced more than 30 lectures and hands-on ...
CHICAGO, US: Aligner therapy has revolutionised the treatment of malocclusion; however, regulators have been unable to keep pace with the rapid market ...
Anterior open bites present orthodontists with a unique challenge: achieving successful and timely treatment while minimizing patient inconvenience. ...
IRVINE, Calif., USA: BIOLASE has announced an upgrade of its WaterLase iPlus 2.0 dental laser to provide dental practitioners a clinical protocol and ...
LONDON, England: After years of hosting leading aligner events across Europe, the European Aligner Society (EAS) will hold its first forum in North America ...
For years the term “orthodontics” conjured images of metal wires, painful brackets and middle school chants of “brace face,” all of ...
Live webinar
Mon. 11 May 2026
12:00 PM EST (New York)
Live webinar
Mon. 11 May 2026
1:00 PM EST (New York)
Live webinar
Tue. 12 May 2026
1:00 PM EST (New York)
Live webinar
Thu. 14 May 2026
12:00 PM EST (New York)
Live webinar
Thu. 14 May 2026
2:00 PM EST (New York)
Prof. Hani Ounsi DDS PhD FICD
Live webinar
Tue. 19 May 2026
11:00 AM EST (New York)
Live webinar
Tue. 19 May 2026
1:00 PM EST (New York)
Prof. Dr. med. dent. Ivo Krejci



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 International / International
International / International
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East



















































To post a reply please login or register