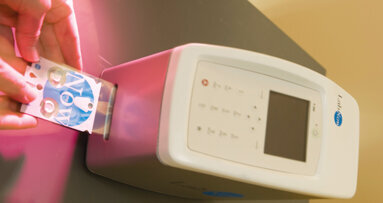
Advanced Laboratory Services, in collaboration with a research team at the University of Southern California, has developed a new test that can assist health care professionals in the early detection, diagnosis and treatment of oral cancers. Advanced Laboratory’s saliva biomarker test measures three specific biomarkers that play a role in cancer development.
As a monitoring tool, the test has the potential to be an essential part of every patient’s annual general-health or dental check-up.
With monitoring of biomarker levels determined by the saliva biomarker test, cancer development in patients can be detected far earlier than previously possible. On average, one person in the United States dies every hour from oral cancer; but it’s not because the cancer is difficult to discover or diagnose. It’s because the cancer is often detected late in its development.
One of the advantages of the saliva biomarker test is that it does not rely on localization of a lesion to detect cancer, and can thus detect oral cancers at treatable stages.
Health care professionals can utilize this test during multiple stages of diagnosis and treatment:
As a yearly screening tool: The test can be used as an annual screening to assess cancer risk-levels in patients 18 and older.
As a prognosis tool: Patients already diagnosed with oral cancer with higher biomarker levels tend to have poorer outcomes, and may require more aggressive treatment.
As a post-treatment monitoring tool: The test can be used to assess whether reoccurrence is likely before a new cancer lesion has developed.
Advanced Laboratory’s test is a simple, noninvasive saliva test that can detect biomarkers across a range of cancers in the oral cavity, including cancers of the tongue, floor of the mouth, cheek lining, gums, palate, salivary glands and tissues that line the mouth and lips.
Health care professionals interested in the effort to find and fight oral cancer can consider use of the test for patients with risk factors for oral cancer — including tobacco and alcohol users and those with persistent viral infections such as the human papilloma virus, or HPV. Patients who have been diagnosed but not yet treated for oral cancer, or who have undergone treatment for oral cancer can also take the test to help with prognosis or detect reoccurrence.
Collection kits consist of a saliva collection tube, instructions, refrigerator pack, requisition form and FedEx mailer. Collection kits are provided to health care professionals free of charge, with no upfront fees or set-up costs. The cost of the test, billed when the sample is received, is $179.99 for all three biomarker levels.
Kits are shipped to Advanced Laboratory Services’ lab in Pennsylvania. The lab is certified by the Clinical Laboratory Improvement Amendments (CLIA) and COLA (formerly Commission on Laboratory Accreditation), and it is HIPAA compliant.
Advanced Laboratory Services Inc. is a state-of-the-art laboratory located just outside of Philadelphia, Pa. With both clinical and research divisions, the company is committed to developing and releasing new, cutting-edge laboratory tests to aid clinical diagnosis and treatment, including its recently launched, exclusive Oral Cancer Saliva Screening test.
(Source: Advanced Laboratory Services)
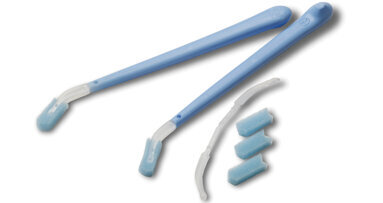
NEW YORK CITY: A new test for oral cancer, which a dentist could perform by simply using a brush to collect cells from a patient’s mouth, is set to be...
The e-Dx endodontic diagnostic tool is available from Jordco. The e-Dx is described as the next generation in endodontic diagnostic instruments. The ...
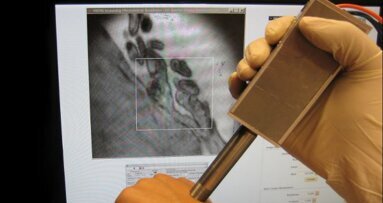
AUSTIN, Texas, USA: In order to reduce the time taken to diagnose oral cancer, researchers have developed the first prototype of a handheld detection device...
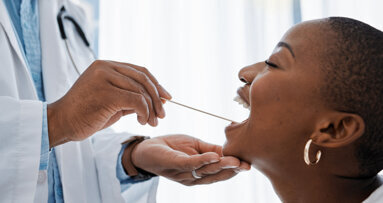
Mark Rutenberg wants you to look for this four-letter word: TEST. Rutenberg is the chairman/CEO of OralCDx, a company that provides the BrushTest, which can...
NEW YORK, US: Oral tumours can cause agonising pain and thus greatly impair the quality of life of patients. Researchers from the New York University ...
HOUSTON, Texas, USA: DentalOne Partners recently joined forces with Forward Science, the manufacturer of OralID. Forward Science’s flagship product, ...
NEW YORK, N.Y., USA: Researchers at UCLA have found that a protein that serves as a suppressor of cancer diminishes in skin and mouth epithelial cells as ...
COLLEGE STATION, Texas, US: A new intra-oral biosensor developed in the US could transform how periodontal disease is detected and monitored. Aiming to help...
SAN FRANCISCO, Calif, USA: Procter & Gamble (P&G) had quite a bit of news to share with the media during the recent ADA annual session, and Dental ...
NEW YORK, N.Y., USA: NYU Oral Cancer Center has been awarded a five-year, $3.1 million grant by the National Cancer Institute (NCI), part of the National ...
Live webinar
Wed. 10 June 2026
11:00 AM EST (New York)
Live webinar
Wed. 10 June 2026
2:00 PM EST (New York)
Nacho Fernández-Baca DDS, MSc
Live webinar
Wed. 10 June 2026
7:00 PM EST (New York)
Live webinar
Thu. 11 June 2026
1:00 PM EST (New York)
Live webinar
Thu. 11 June 2026
2:00 PM EST (New York)
Prof. Hani Ounsi DDS PhD FICD
Live webinar
Fri. 12 June 2026
8:00 AM EST (New York)
Dipl.-Hdl. Joachim Brandes
Live webinar
Mon. 15 June 2026
11:00 AM EST (New York)
Dr. Cristian Scognamiglio, Dr. Alessandro Perucchi



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 International / International
International / International
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East
















































To post a reply please login or register