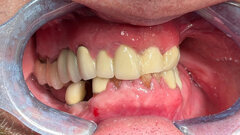
NEW YORK/LEIPZIG, Germany: The US Food and Drug Administration have has given clearance for Genesis, a new implant system developed by Keystone Dental in Burlington, Massachusetts, in the United States. The new system is said to improve the aesthetic outcome of implant procedures through a pink implant collar called AnaTite. It also offers a new nano-surface that mimics the structure of bone and improves osseointegration, the company says.
Keystone offers a variety of dental products including the PRIMA implant which the company acquired from an aquisition of Lifecore Biomedical in 2008. The FDA clearance will give the company the right to market Genesis immediately in the combined North American markets. According to the company, it is expected to be launched in Europe in February 2011.
NEW YORK, USA/LEIPZIG, Germany: Dentists in the USA now have access to the new OsseoSpeed TX line from Astra Tech. The Sweden-based manufacturer launched ...
CHARLOTTE, N.C., US: Dentsply Sirona and Siemens Healthineers have announced that their dental MRI system, MAGNETOM Free.Max Dental Edition, has received ...
MIAMI, Fla., USA: Neocis, a supplier of robot-assisted dental implant surgery, has announced it has received 510(k) clearance from the U.S. Food and Drug ...
LANSDALE, Pa., USA: X-Nav Technologies — a medical device company that develops surgical products intended to advance patient care while improving doctor ...
HOUSTON, Texas, USA: OrthoAccel Technologies, a manufacturer of accelerated orthodontics technology, has received FDA clearance for its newest accelerated ...
JUPITER, Fla., USA: Southern Implants North America (SINA) has received FDA 510(k) clearance for its innovative internal hex dental implant system, PROVATA,...
Nuventus, a privately held Swiss dental implant company, has announced the official commercial launch of the Nuventus NV.C Implant System in the United ...
BOSTON, US: Overjet, a dental start-up that develops software powered by artificial intelligence (AI), recently announced that it has received 510(k) ...
Sonendo announced recently that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for the next generation of its GentleWave ...
FORT LAUDERDALE, Fla., US: Swiss dental implant specialist Patent Medical has been named “Dental Implants Manufacturer of the Year in Europe” by MedTech...
Live webinar
Wed. 27 May 2026
8:00 AM EST (New York)
Live webinar
Thu. 28 May 2026
12:00 PM EST (New York)
Live webinar
Mon. 1 June 2026
1:00 PM EST (New York)
Live webinar
Wed. 3 June 2026
1:00 PM EST (New York)
Live webinar
Thu. 4 June 2026
2:00 PM EST (New York)
Live webinar
Mon. 8 June 2026
12:00 PM EST (New York)
Live webinar
Mon. 8 June 2026
1:00 PM EST (New York)
Dr. Anthony Mak B.D.S, Prof. Marleen Peumans



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 International / International
International / International
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East



















































To post a reply please login or register