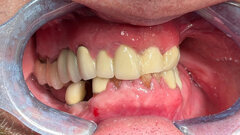
As clinical dental hygienists, we want our patients to be as healthy as possible. This means having the skills and knowledge to treat them as effectively as possible. When a patient presents with periodontal disease, scaling and root planing with or without osseous surgery are the modalities for treating this condition.
However, what about the cases that do not respond well to these treatments? It is important for us to keep ourselves aware new treatment developments that may provide some more hope for these patients.
This could involve looking outside the dental field. I learned about this new treatment for osteoporosis from a patient of mine who is an orthopedic physician.
We are all aware of the bisphosphonate drugs for treating osteoporosis and the potential for complications they bring. A new drug for osteoporosis has been shown to regenerate bone. Teriparatide, marketed under the name Forteo, is a parathyroid hormone injection taken once a day.
Forteo is used to treat osteoporosis in cases that do not respond to traditional treatments. It has just recently been introduced and is still very expensive for the patient, so bisphosphonates are still the first choice for the treatment of osteoporosis.
Forteo is the portion of human parathyroid hormone (PTH) amino acid sequence 1 through 34 of the complete 84 amino acid molecule. PTH is the primary regulator of calcium and phosphate metabolism in bone and the kidneys. PTH increases the serum calcium partially caused by bone resorption. Thus, chronically elevated levels of PTH can lead to depleted bone stores, however, intermittent exposure by a daily 20-microgram injection will activate osteoblasts more than osteoclasts. Daily injections of teriparatide have a net effect of stimulating new bone formation, leading to increased bone mineral density.[2–4] Teriparatide was approved by the FDA on November 26, 2002, for the treatment of osteoporosis in men and post-menopausal women who are at high risk for fractures. The drug is also approved to increase bone mass in men with primary or hypogonadal osteoporosis who are at high risk for fractures. It is the first, and to date only, FDA-approved agent for the treatment of osteoporosis that stimulates new bone formation.[5]
When I heard about Forteo I was interested to see if any studies had been done on the regeneration of alveolar bone. Could you imagine: A drug that could regenerate bone in the jaw could potentially cure periodontal disease? How exciting for dentistry!
It turns out there is only one study, conducted at the University of Michigan, “Teriparatide and Osseous Regeneration in the Oral Cavity.”[1] The study had 40 participants with severe, chronic periodontitis who had undergone periodontal surgery and received daily injections of teriparatide or placebo, with oral calcium and vitamin D supplementation for six weeks. The patients were followed for one year.
Primary outcome was radiographic linear measurement of alveolar bone level. The mean linear gain in bone at one year in the teriparatide patients was 29 percent versus 3 percent in the placebo patients. Reduction of periodontal probing depth was 33 percent versus 20 percent and a gain in clinical attachment level of 22 percent versus 7 percent. Teriparatide, as compared with placebo, was associated with improved clinical outcomes, greater resolution of alveolar bone defects and accelerated osseous wound healing in the oral cavity.
No adverse reactions were experienced in this study, but the sample population was very small. Teriparatide was shown in animal studies to increase the risk of osteosarcoma. This is the biggest risk of taking Forteo, and it is a pretty serious one. Patients with a history of Paget’s Disease of bone or unexplained elevations of serum alkaline phosphate, open epiphysis or prior radiation therapy involving the skeleton should not take Forteo. Patients taking Forteo should not smoke or drink alcohol, and the most commonly reported side effects are nausea, leg cramps and dizziness.
Forteo is definitely a promising method for treating periodontal disease, but the risk of bone cancer is definitely something patients and practitioners need to consider. We must remember as clinicians that our traditional methods have had success and should be utilized first, and Forteo can be used in the cases that do not respond to scaling and root planing or periodontal surgery.
Forteo certainly will change how we view periodontal cases, but at this time it is a risky treatment and has not been studied enough with periodontal patients to say for sure that it will eradicate periodontal disease.
The introduction of this drug and the potential for new modalities of treatment do offer some promise for our patients, but we need more information.
More studies need to be done and more information gathered before we can decide if Forteo is an appropriate treatment for resistant periodontal cases. Hopefully, we will be hearing more about this drug and how it relates to dentistry in the future.
In the meantime, we will continue to provide our periodontal patients the best care we can using our traditional methods. The future is bright for dentistry!
Note: This article was originally published in Hygiene Tribune U.S. Edition, Vol. 4 No. 9, September 2011. A complete list of references is available from the publisher.
About the author
Jeanne R. Bosecker, BSN, RDH, is a private-practice dental hygienist living in the Chicago suburbs. After graduating from nursing school, she found her true passion in dentistry after working as a dental assistant for several years. Since graduating with her dental hygiene degree, she has worked for three small general dentistry practices. You may contact her at jeannebosecker@comcast.net.
NORTHBOROUGH, Mass., USA: Solmetex announced today (May 19, 2020) that it has received additional patents for its NXT Amalgam Separator System, including ...
After years of schooling, thousands of dollars in tuition, hours upon hours of clinics and exams, and tests and on and on, finally you entered the working ...
You’ve been learning about all the new products and risk assessment tools in lectures, articles in magazines and on the Internet. Saliva tests have ...
When consumers fire up their web browsers, the vast majority of them start at a search engine. According to research from Pew Internet, 93 percent of online...
Summer comes early to Colonial Williamsburg. Poppies, our nation’s brilliant symbol of remembrance, are in full bloom by Memorial Day. I just love the...
We hear about it a lot these days — dental practices getting squeezed by lower consumer spending. The result is less treatment acceptance and lower ...
NASHVILLE, Tenn., USA: Seven out of 10 American adults have crooked and misaligned teeth and have wanted to straighten them for years, but haven’t ...
CHICAGO, Ill., USA: The American Association of Endodontists (AAE) has developed and launched a new website that offers more content and easier navigation ...
NEW YORK, NY, USA: A facial exercise programme involving devices used by dentists has shown prospects for the rehabilitation of facial nerve damage. ...
CHICAGO, Ill., USA: Convergent Dental, developer of Solea, a computer-aided, CO2 all-tissue dental laser system, is announcing the launch of Solea SW 3.1.1 ...
Live webinar
Wed. 3 June 2026
1:00 PM EST (New York)
Live webinar
Thu. 4 June 2026
2:00 PM EST (New York)
Live webinar
Mon. 8 June 2026
12:00 PM EST (New York)
Live webinar
Mon. 8 June 2026
1:00 PM EST (New York)
Dr. Anthony Mak B.D.S, Prof. Marleen Peumans
Live webinar
Mon. 8 June 2026
2:00 PM EST (New York)
Live webinar
Wed. 10 June 2026
11:00 AM EST (New York)
Live webinar
Wed. 10 June 2026
2:00 PM EST (New York)
Nacho Fernández-Baca DDS, MSc



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 International / International
International / International
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East














































To post a reply please login or register