It is estimated that 6 percent of the American population — 18 million people — are congenitally missing a maxillary lateral incisor. To address this need, DMX Implant Corp., the dental implant division of Dentatus Ltd., has created a unique narrow-body implant called the ANEW Implant System. ANEW is the only narrow diameter implant that accepts a screw-retained abutment. This advantage affords prosthetic options unlike other narrow diameter implants.
embedImagecenter("Imagecenter_1_394",394, "large");
The high success rate of narrow-body implants has expanded treatment options for both dentist and patient. Primarily, narrow-body implants can be placed into anatomically challenging areas that would be contraindicated for standard diameter implants without site modification procedures, such as bone grafting and orthodontics.
These procedures increase treatment time, cost to the patient and morbidity. This can deter the patient from dental implant therapy, thereby subjecting the patient to limiting their treatment plan to less definitive options such as “flipper” appliances, removable partial dentures and “bonded” and conventional bridges.
In 2001, in conjunction with the NYU Department of Dentistry, DMX established a specific prosthetic protocol. In 2004, the FDA approved ANEW Implants for “long-term use or any length of time as determined by the health-care provider.” The low profile 3 mm head accommodates divergent angles, offering natural looking esthetics.
The non-hygroscopic screw-cap abutment facilitates fabrication of a fixed transitional restoration at the time of implant placement, thereby providing the patient with an immediate, predictable and cosmetic result. During the healing period, the restoration contours can be easily modified to the contours of the tissue architecture, thereby eliminating a final “black triangle” result.
ANEW narrow-diameter implants are minimally invasive and designed to fit into narrow spaces with implant diameters of 1.8, 2.2 and 2.4 mm respectively. The ANEW tapered, one-piece implant design eliminates microgap related crestal bone loss, facilitates one-stage surgery, provides immediate restoration and is more conducive to flapless implant placement.
Additionally, utilizing a minimally invasive flapless procedure with an immediate restoration eliminates many postoperative challenges as well as reduces total treatment time.
ANEW narrow diameter implants have been tested with university-based research from the around the world. In 2007, Dr. Stuart Froum and his colleagues from the New York University Department of Implant Dentistry published a study in the International Journal of Perio and Restorative Dentistry stating “40 Anew Implants in patients for 1 to 5 years postloading. No implant failures were reported, yielding a 100% survival rating.”
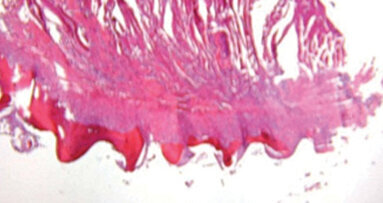
In 2005, the Journal of Oral and Maxillofacial Implants published Dr. Michael Rohrer’s histology study on Dentatus implants. Dr. Rohrer determined that the percentage of bone in contact with the body of Dentatus implants in “the same range and sometimes higher than what is usually seen with conventional implants.”
These results support well-known literature about implant design and materials in the following ways: ANEW narrow-body dental implants are composed of Grade V titanium alloy; the threaded portion of the implant is mechanically roughened to increase surface area and maximize the bone-implant interface; and the tapered design better facilitates implant placement, promotes initial implant stability and better distributes occlusal loads along the body of the implant. Predictably, ANEW implants have been placed in various places within the mouth with high success.
Case study
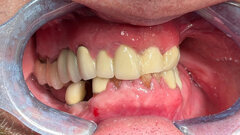
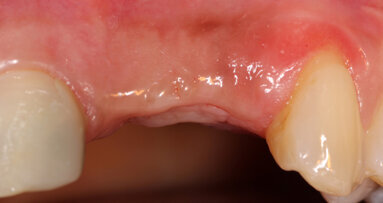
A 15-year-old girl and her father came to the office for diagnosis and treatment planning as her orthodontic treatment was coming to an end. She presented with congenitally missing lateral incisors. Her orthodontic treatment had provided appropriate root separation of the cuspids and centrals as well as good esthetics during treatment. This was accomplished by having a prosthetic tooth, #7, suspended from the archwire and retention of the upper left deciduous lateral incisor throughout the entire treatment course (Figs. 1–3).
The treatment plan accepted was to proceed with the completion of the orthodontic treatment and debracketing (Fig. 4) that same day, extract the upper left deciduous lateral incisor and then place ANEW implants in the lateral incisor positions of #7 and #10.
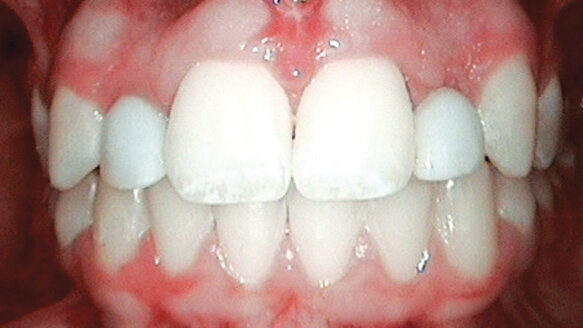
Once the ANEW implants were placed, an immediate fixed provisional crown would be fabricated on each implant. They would then be held in static occlusion as part of the orthodontic retention as well as to help provide initial stability for the ANEW implants during osseointegration (Figs. 5, 6).
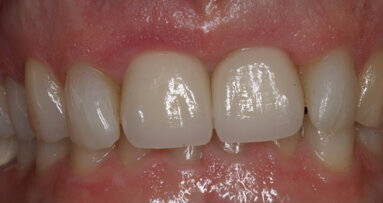
It was clearly understood that as the still growing patient would continue to mature, the provisional crowns would need to be removed and revised and/or remade in order to properly form the papillae and modify the incisal length.
This would easily be accomplished with the ANEW screw-retained abutment and provisional crown possibilities (Figs. 7–9). The final restorations supported by the ANEW Implants will be fabricated when the growth of the premaxilla is complete in 4 to 5 years at age 19 and 20.
Note: This article was originally published in Dental Tribune U.S. Edition, Vol. 6 No. 15, September 2011.
About the authors
Robert M. D’Orazio, DDS, ABOI/ID, is a graduate of the University of Detroit, School of Dentistry. He currently maintains a referral-based implant dental practice located in Sterling Heights, Mich.
Mark A. Iacobelli, DDS, FAGD, FICD, MIIF, is a graduate of Case Western Reserve School of Dentistry. He has been in private practice since June 1982 and holds licenses and sedation permits in the states of Ohio and Florida. He lectures the DMX Implant Corporation, the dental implant division of Dentatus Ltd.
After years of teaching at endodontic programs around the country, I can say with strong conviction that the process of critical thinking has not been ...
LOUISVILLE, Ky., USA: Tooth loss can lead to poor nutrition and is linked to systemic diseases such as diabetes and coronary heart disease. Missing teeth ...
A new review article by US researchers explores the inequities and biases in oral healthcare and highlights the potential of artificial intelligence (AI) in...
Most dentists will say, “I have good systems.” However, are they the right systems? Inefficient and outdated systems create a no-win situation ...
Mini-implant dentistry has long been a controversial topic, which has steered many dental practitioners across the world away from reaping the multitude of ...
This article is by Arun K. Garg, DMD; Gregori M. Kurtzman, DDS; Renato Rossi Jr., DMD, MSc, PhD; and Maria del Pilar Rios. DMD. MSc. PhD
A medically and periodontally stable 50-year-old woman presented with failing #8 and #9 teeth that exhibit asymmetry, lack of interdental papilla and a ...
SACRAMENTO, Calif., USA: An article titled “Lateral Wall Sinus Floor Elevation for Implant Placement: Revisiting Fundamentals and the Surgical ...
Live webinar
Wed. 3 June 2026
1:00 PM EST (New York)
Live webinar
Thu. 4 June 2026
2:00 PM EST (New York)
Live webinar
Mon. 8 June 2026
12:00 PM EST (New York)
Live webinar
Mon. 8 June 2026
1:00 PM EST (New York)
Dr. Anthony Mak B.D.S, Prof. Marleen Peumans
Live webinar
Mon. 8 June 2026
2:00 PM EST (New York)
Live webinar
Wed. 10 June 2026
11:00 AM EST (New York)
Live webinar
Wed. 10 June 2026
2:00 PM EST (New York)
Nacho Fernández-Baca DDS, MSc



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 International / International
International / International
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East












































To post a reply please login or register