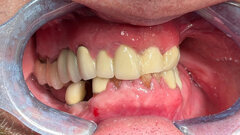
NEW YORK, NY, USA: Thousands of dental offices in the United States and in countries around the world have experienced the quality, proven safety, and convenience of the Aribex NOMAD handheld X-ray system. Because it is lightweight and rechargeable, the device has also been used to diagnose thousands of individuals in remote areas far from a regular dental operatory.
NOMAD has also helped dental professionals in treating special needs patients such as the elderly, the handicapped, and those under sedation. And, thanks to the loving hearts of countless volunteer professionals, it has been proven to be of significant value for dental missions in clinics, orphanages and schools throughout the United States and in isolated villages in developing countries.
Even after the FDA clears an X-ray device as safe and effective, each state radiation control section must approve the device for use in its state. While most have approved handheld X-ray devices, some still have not. Unfortunately, the same NOMAD X-ray device used to help a child in Mozambique is not available in states such as New Hampshire, Kentucky, Maryland, Minnesota, Michigan, Delaware, or in Canada. Then there are a few states that severely restrict the use of handheld X-ray to specific circumstances, or apply burdensome requirements, which ultimately discourage use.
“Dentists in restrictive states and in Canada need to let regulators know that they want to provide the higher level of care available through handheld X-ray,” said Ken Kaufman, president of Aribex. “NOMAD has been tested extensively and found to be as safe as or even safer than the conventional units dentists are using now. State dental associations acknowledge that access to care is a major issue in their state, and the American Dental Association has worked to raise awareness. Still, some state regulators haven’t gotten the message that the NOMAD handheld X-ray is a huge part of the solution. Dentists need to let regulators know what they want.”
The FDA recently announced an investigation into foreign-made handheld X-ray devices being sold into the United States over the Internet. None of these units have been approved for use in any state.
“Aribex has spent years in painstakingly obtaining state approvals,” said Kaufman. “We’re concerned that inferior units being sold illegally will muddy the waters for regulators considering our device in their state. And that will mean additional roadblocks for access to care.”
(Source: Aribex)
CHICAGO, IL, USA: “Access to care is really what the message is,” said Ken Kaufman, president and chief financial officer of Aribex, supplier of...
Recognitions continue to accumulate for the advances in radiography brought about by the NOMAD handheld X-ray system. On Jan. 19, D. Clark Turner, PhD, ...
Aribex, which describes itself as the worldwide leader in handheld X-ray technologies, has completed production of its 10,000th NOMAD handheld X-ray system....
Aribex, a worldwide leader in handheld X-ray technologies, announced Sept. 29 that its NOMAD handheld X-ray has been named as a runner-up in the medical ...
The NOMAD Pro Handheld X-ray System by Aribex has become the choice of thousands of dental practitioners across the country for its incredible portability, ...
OREM, UTAH, USA: Aribex, supplier of handheld X-ray technologies and manufacturer of the NOMAD Pro handheld X-ray system, recently announced 2011 ...
OREM, UTAH, USA: Aribex, a manufacturer of handheld X-ray technology, has qualified for up to $350,000 in financial incentives from the state of Utah for ...
The American Association of Oral and Maxillofacial Surgeons will return to sunny Honolulu at the Hawaiian Convention Center this Sept. 8-13.
LAS VEGAS/NEW YORK, USA: We’ve all heard the saying: ‘What happens in Vegas, stays in Vegas.’ Except this time, you’re not going to ...
If you haven’t yet registered for the American Academy of Implant Dentistry’s Virtual Experience, it’s not too late. The annual meeting, which started...
Live webinar
Wed. 3 June 2026
1:00 PM EST (New York)
Live webinar
Thu. 4 June 2026
2:00 PM EST (New York)
Live webinar
Mon. 8 June 2026
12:00 PM EST (New York)
Live webinar
Mon. 8 June 2026
1:00 PM EST (New York)
Dr. Anthony Mak B.D.S, Prof. Marleen Peumans
Live webinar
Mon. 8 June 2026
2:00 PM EST (New York)
Live webinar
Wed. 10 June 2026
11:00 AM EST (New York)
Live webinar
Wed. 10 June 2026
2:00 PM EST (New York)
Nacho Fernández-Baca DDS, MSc



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 International / International
International / International
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East


















































To post a reply please login or register