Recent studies provide increasing evidence that untreated periodontal inflammation is linked to cognitive decline and Alzheimer’s disease, and that treating periodontitis may reduce or delay risk of this disease. About one out of every nine Americans have some form of Alzheimer’s disease, according to the Alzheimer’s Society. It is the worst type of dementia, involving the most cognitive decline and memory loss.
There is no known cure, and the magnitude and severity of this fatal disease mandates that research continues to explore every avenue that might lead to one.
Research has already found Porphyromonas gingivalis (P. gingivalis) in brains of Alzheimer’s patients. P. gingivalis is a bacterium associated with chronic periodontitis. Researchers propose that when these bacteria reach the brain, an immune response is stimulated in the brain to release proteins that kill the bacteria, but also cause broader destruction.
Periodontal disease bacteria enter bloodstream, brain
A study by professor Stjohn Crean and Dr. Sim Singhrao at the University of Central Lancashire (UCLan) School of Medicine and Dentistry in England involved the examination of brain tissue samples of 10 deceased people with Alzheimer’s and 10 people without it.
It was found that bacteria found in chronic periodontal disease were present in the brains of four of the 10 people with Alzheimer’s but in none of the 10 without it. Researchers theorized that chronic periodontal disease bacteria, P. gingivalis, enter the bloodstream and brain, prompting an immune system response, which over time is thought to contribute to cognitive decline and Alzheimer’s disease.
Amyloid proteins trap microbes
Recently, there was a study done at Massachusetts General Hospital, funded by the National Institutes of Health. Dr. R. Tanzi’s research with live mice showed that beta amyloid proteins in live mice trapped microbes — but the microbes remained free in live mice without the amyloid proteins.
Results indicated that with no amyloid to trap the infections, infection spread rapidly; but with the amyloid, infection was stopped. However, the study revealed that too much beta amyloid protein can over clump-down on itself, without trapping microbes — indicating that efficacy requires a delicate balance.
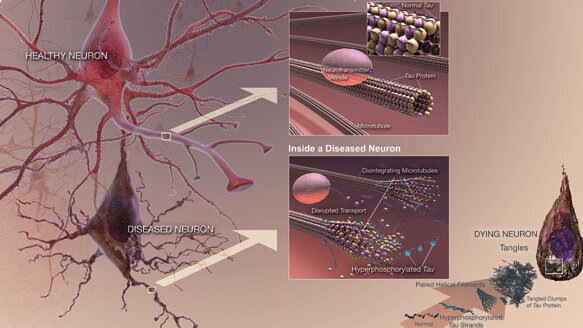
Tanzi further theorized that genes play a large roll, specifically ApoE2 and ApoE4. The gene ApoE2 enabled the brain to clean out the beta amyloid after it killed the microbes, leading to minimal cognitive decline in old age. The gene ApoE4, however, was inefficient at enabling such a “cleanup,” and the risk of developing Alzheimer’s disease was much greater.
All blood vessels are anastomosed in the human body. Therefore, all have connecting relationships and can transmit an inflammatory by-product easily. As mentioned, numerous articles allude to this connection with regard to periodontal tissue circulating to the heart and brain.
At last year’s induction of fellowships for the International Academy for Dental Facial Esthetics in New York, Dr. Ray Williams, former dean of Stony Brook Dental School and a highly regarded researcher, presented a detailed overview of the latest findings on periodontal vascularity. Williams listed numerous publications showing a direct vascularity relationship between the periodontia and the heart. However, Williams also listed an equal amount of publications that infer no direct connection between periodontia and the heart.
Ability to self-maintain oral health can diminish with age
A key area that needs to be addressed is the challenge of self-maintenance of periodontal disease as people’s physical abilities diminish with age.
When I had the privilege to be involved with several senior-care facilities, it was obvious that a large number of residents were unable to maintain their oral health at a desired level. Some residents with dentures would misplace or lose a prosthesis or even borrow one from another resident.
Earlier this year, researchers at Harvard University conducted a study hypothesizing that infections such as chronic periodontitis produce reactions that leave debris in the brain that later contribute to Alzheimer’s disease.
The study found that in the subjects involved, bacteria passed through the blood-brain barrier, prompting the brain’s defense mechanism to release the “sticky” beta amyloid protein. This protein entrapped and killed the bacteria, but what remained was plaque, termed “the hallmark of Alzheimer’s.” This plaque signaled another brain defense mechanism — a release of a tangle of tau proteins that not only killed the plaque, but also the brain’s nerve cells, leading to more and more brain inflammation — and resulting in continued cognitive decline.
Vicious, repetitive cycle
The vicious, repetitive cycle leads to further and further destruction. However, not every brain was adversely affected because of the periodontal bacteria.
Researchers concluded that genetically, some brains (ApoE2) can clean out the beta amyloid, get rid of the plaque and lower the risk for Alzheimer’s — but other brains (ApoE4) can’t, correlating to a higher risk of getting the disease.
Alzheimer’s disease, with its cognitive decline and memory loss, is destructive not only to the patient, but to all of the patient’s loved ones. Understanding and addressing periodontal vascularity may well be a major pathway toward better and longer functioning of the brain.
Perhaps going to the periodontist is the most expeditious, economical and easiest pathway to slowing cognitive decline and onset of Alzheimer’s disease.
This article was published in Dental Tribune U.S. Edition, Vol. 11 No. 10, October 2016 issue.
NEW YORK, U.S.: Older adults with more harmful than healthy bacteria in their periodontal tissue are more likely to have evidence for amyloid beta— a key ...
NEW YORK, N.Y., USA: The American Academy of Periodontology (AAP) supports the scientific statement of the American Heart Association (AHA), ...
CHICAGO, Ill., USA: Despite provocative reporting and sensational headlines, the American Association of Endodontists says there is no evidence that ...
MINNEAPOLIS, US: Since the election of Donald Trump as US president in 2024, the question of water fluoridation in the country has become increasingly ...
NEW YORK, N.Y., USA: A Danish–American study has proven that incidences of Alzheimer's disease can be traced back to chronic gum inflammation. ...
NEW YORK, NY, USA: NYU dental researchers have found the first long-term evidence that periodontal (gum) disease may increase the risk of cognitive ...
ROCHESTER, N.Y., US: In what is thought to be the first study to directly link prenatal stress hormones with primary tooth eruption, researchers in the US ...
CLEVELAND, OH, USA: Researchers at the Department of Periodontics at Case Western Reserve University School of Dental Medicine reported the first documented...
SEATTLE, US: Ageing greatly affects overall health and is the greatest risk factor for cardiovascular disease, Alzheimer’s disease and periodontal ...
MORGANVILLE, N.J., USA: November is American Diabetes Month, and the New Jersey Society of Periodontists (NJSP) would like patients and caregivers alike to ...
Live webinar
Tue. 26 May 2026
12:00 PM EST (New York)
Live webinar
Tue. 26 May 2026
2:00 PM EST (New York)
Live webinar
Wed. 27 May 2026
8:00 AM EST (New York)
Live webinar
Thu. 28 May 2026
12:00 PM EST (New York)
Dr. Anthony Viazis DDS, MS. Inventor & Founder of Fastbraces®
Live webinar
Mon. 1 June 2026
1:00 PM EST (New York)



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 International / International
International / International
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East















































To post a reply please login or register