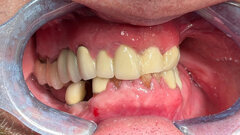
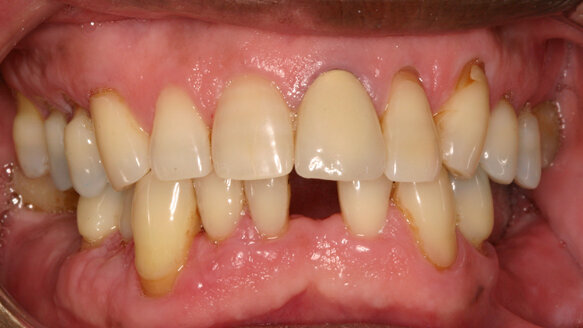
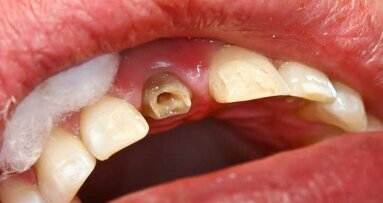
The use and acceptability of implants today is considered both routine and highly predictable. With that, people more than ever before are considering the replacement of missing teeth by this method. Unfortunately, until now, a segment of this population has not been viewed as a viable candidate, particularly patients with compromised situations having to do with limited interdental spaces, advanced bone loss, convergent roots, age and financial constraints.
Anew Implants (Dentatus, USA, New York) are “ideally designed for the compromised implant site; these 1.8 mm, 2.2 mm and 2.4 mm diameter implants offer a fixed permanent tooth replacement option for patients who otherwise would not be able to have implants placed and restored,” wrote Paul Petrungaro in “Clinical Briefs” in the publication Inside Dentistry, No. 3/2006.
The Anew Implant design offers practitioners a unique chairside, screw-retained restoration that fits where conventional diameter implant systems do not. The recommended surgical techniques allow for minimally invasive placement (no flaps) and immediate loading. This eliminates most postoperative challenges and dramatically reduces the total time in treatment.
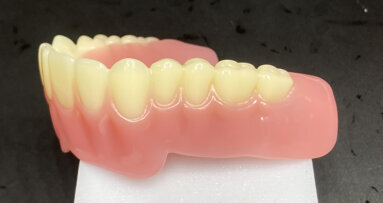
The economical prosthetic components designed for all sizes and lengths of Anew Implants are used for interim restorations and laboratory procedures. The advantage is immediate fixed temporization at implant placement without the need for removable appliances during the osseointegration interval.
Rather than having a recess within the implant, the screw is formed on the coronal aspect of the implant. The crown is stabilized on the square platform and secured with a resin screw-cap that is recessed within the abutment, which allows for retrieval without excess force or cross-thread damage to implants. Crowned tissue contacts can be incrementally added for guiding tissue emergence profiles and creating esthetic papillae, preventing black-hole syndromes between teeth.
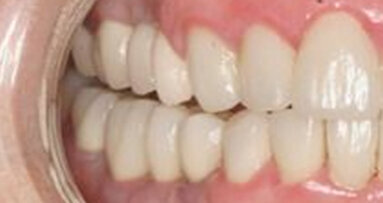
The narrow, polished platform and a short external screw abutment of Anew Implants help to create exceptional esthetics with sculpted tissue forms for tooth emergence profiles. The non-hygroscopic screw-cap abutment allows implants to be easily monitored, altered and adapted to a permanent restoration.
With Anew Implants, patients’ quality of life and ability to maintain a normal lifestyle during the course of treatment is significantly enhanced. These implants can provide implant therapy to a much larger segment of the population, one that was previously neglected.
Anew Implants, made of Grade 5 Ti-alloy, have an etched surface for improved stability and osseointegration and are packaged pre-sterilized. In 2004, they were granted FDA approval. The restorative protocol was developed in conjunction with the Department of Implant Dentistry at New York University College of Dentistry. Numerous published clinical and histological studies of the Anew Implant report excellent bone adaptation and high survival levels, in addition to 100 percent patient satisfaction.
embedImagecenter("Imagecenter_1_187",187, "large");
More information is available from Dentatus.
The unceasing innovations and iterations in dental technologies and materials have taken the sophistication of treatment modalities to a historic level. ...
Nassau, Bahamas-based Paradise Dental Institute formally launches this July with a live-surgery implant training course followed by a working conference. ...
Given the great success and ease of use of the Champions one-piece implant system, the question of why the development of a two-piece implant system was ...
In this ever-evolving age of dental technology, the practice area of implant site development has benefitted from a technologic innovation and a new era of ...
How many times have you seen a case during an examination where the soft-tissue collar was less than you would like to have? How many times has this ...
Implant site preservation is an important component of diagnosis and treatment planning. Through CAD (computer aided design), prosthesis can be designed ...
ROCK HILL, S.C., US: 3D Systems has announced the US commercial launch of its NextDent Jetted Denture Solution. The denture system utilises multiple ...
Dentsply Sirona is ready for AO 2024 with a full line-up for our Inspiration TALKS at the Corporate Forum and the latest product and technology solutions ...
Dental Tribune interviewed Dr. Paresh B. Patel, one of the featured speakers at the ICOI Winter Implant Symposium, which took place Feb. 12-14 at the Trump ...
Implant dentistry is an expanding field. Ensure your skills are strengthened and that you can manage every appropriate phase of your implant care through a ...
Live webinar
Wed. 3 June 2026
1:00 PM EST (New York)
Live webinar
Thu. 4 June 2026
2:00 PM EST (New York)
Live webinar
Mon. 8 June 2026
12:00 PM EST (New York)
Live webinar
Mon. 8 June 2026
1:00 PM EST (New York)
Dr. Anthony Mak B.D.S, Prof. Marleen Peumans
Live webinar
Mon. 8 June 2026
2:00 PM EST (New York)
Live webinar
Wed. 10 June 2026
11:00 AM EST (New York)
Live webinar
Wed. 10 June 2026
2:00 PM EST (New York)
Nacho Fernández-Baca DDS, MSc



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 International / International
International / International
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East













































To post a reply please login or register